|
ANORO is not indicated for the treatment of asthma. use of a long-acting beta 2-adrenergic agonist (LABA), including vilanterol, one of the active ingredients in ANORO, without an inhaled corticosteroid (ICS), in patients with asthma.patients with severe hypersensitivity to milk proteins or who have demonstrated hypersensitivity to umeclidinium, vilanterol, or any of the excipients. 
She said that a combination of a LABA and LAMA is restricted to patients with a forced expiratory volume in one second (FEV1) of more than 50% who cannot be prescribed an inhaled corticosteroid, not less than 50%. This article was amended on 7 October 2014 to correct an error in Anna Murphy’s quote.The most common side effects of Anoro Ellipta are upper respiratory tract infection, urinary tract infection, inflammation of the throat, sinuses or nose, headache, cough, mouth and throat pain, constipation and dry mouth. However, the product did not lead to any clear improvement in breathlessness or quality of life. The next stage is the addition of an inhaled corticosteroid (ICS) to LABA therapy and if exacerbations continue, triple therapy with a LAMA, LABA and ICS can be considered.Īnoro Ellipta was studied in eight phase III clinical trials (n=6,835) and it was found to significantly improve lung function compared with either vilanterol, umeclidinium or tiotropium alone - a LAMA commonly used as maintenance therapy for COPD. If exacerbations persist then patients can be upped to a long-acting beta 2 agonist (LABA) or muscarinic antagonist (LAMA), such as tiotropium. Initial treatment of COPD is with a short-acting beta 2 agonist (SABA) such as salbutamol, or a short-acting muscarinic antagonist (SAMA). 
The inhaler is designed to be used once daily at the same time each day.ĬOPD is the name for a collection of diseases that cause the airways of the lungs to narrow and become less elastic, leading to symptoms such as breathlessness and cough. It is available in an Ellipta inhaler device, which is a simple device, and in practice patients may find it easier to use than alternative dry powder inhalers.” Murphy says it is essential that prescribers ensure that the patient has COPD rather than asthma or overlap disease - when patients have symptoms of both asthma and COPD - as these patients should always be given a LABA with an inhaled corticosteroid.īut there are some advantages of the product, highlighted Toby Capstick, lead respiratory pharmacist at Leeds Teaching Hospitals NHS Trust: “It is a combination LABA/LAMA inhaler, which may improve adherence compared to using separate LABA and LAMAs. She believes such a combination bronchodilator is appropriate for people with breathlessness that persists after referral to pulmonary rehabilitation, receiving smoking cessation advice and having had optimisation of their other treatments. It is restricted to a small cohort of patients with a forced expiratory volume in one second (FEV1) of more than 50% who cannot be prescribed an inhaled corticosteroid.” “The recommended place for a combination of a LABA and LAMA is extremely limited in the current COPD guideline from the National Institute for Health and Care Excellence. It is unclear where these products fit into the management of COPD, says Anna Murphy, consultant respiratory pharmacist at University Hospitals of Leicester NHS Trust. Another similar product, Duaklir (aclidinium bromide plus formoterol fumarate), is due to be launched by Almirall in December 2014. 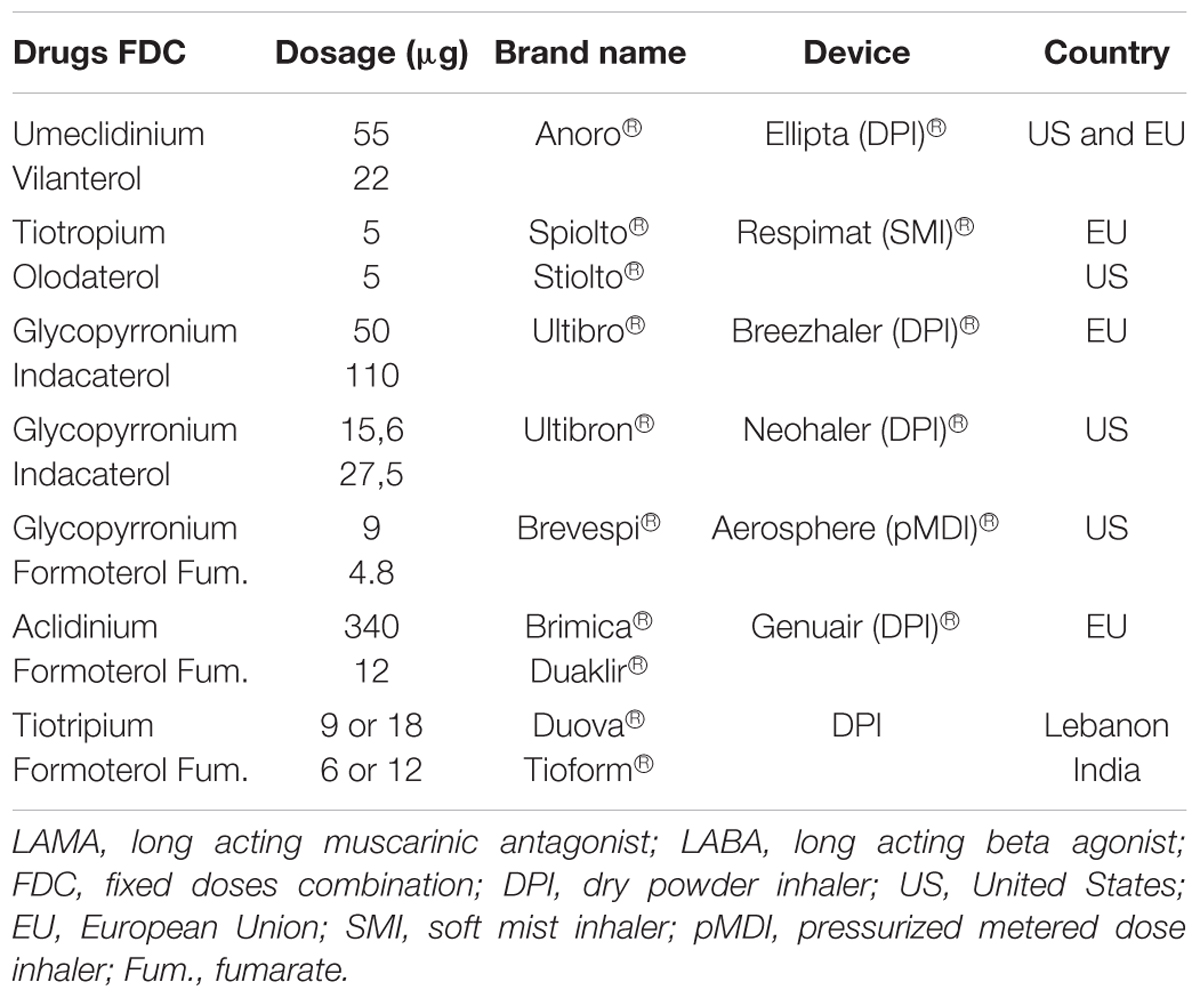
The first product to combine a LABA and a LAMA, Ultibro (Novartis’s indacaterol plus glycopyrronium), was granted a licence in September 2013, but it is yet to be made available in the UK. The product is licensed as a maintenance treatment to relieve symptoms in adults with COPD, but it has been rejected for use within NHS Scotland by the Scottish Medicines Consortium.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
